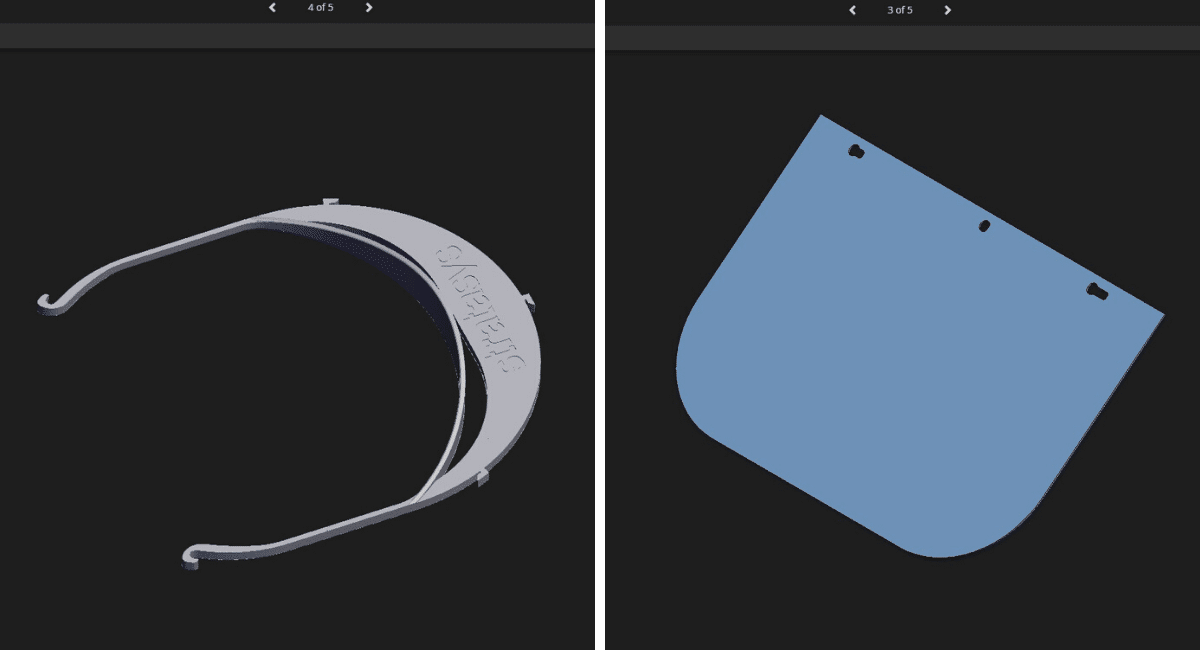
Stratasys is responding to the COVID-19 pandemic by ramping up the production of 3D-printed personal protection equipment. In the United States, having set an initial goal of producing 5,000 face shields at no cost to recipients, the company expects to ship 120,000 by the end of April. This includes a 3D-printed frame and clear plastic shield that covers the entire face. The company will be scaling to a faster production rate. Any 3D printing shop or company using a 3D Printer that wishes to help print plastic frames can fill out an online form to be invited to join the effort. The company is also posting the full face shield printing and assembly instructions on its COVID-19 response page. Célia Sampol, Editor in chief at our sister platform MedicalExpo magazine talked to Eric Bredin, Vice President Marketing EMEA at Stratasys.
This interview was initially published in MedicalExpo magazine.
Célia Sampol: Could you tell us more about this initiative to 3D print facial screens to give to hospitals?
Eric Bredin: This initiative started in the United States. We tried to identify through the healthcare network we have where the needs were and what we could bring to the effort that was most relevant with 3D printing technology. The notion of a conventional tissue or paper-based face mask is not very realistic for us because it’s not really applicable for our technology or materials, it’s not where we can add value and it usually requires huge volumes for disposable masks which 3D can’t do compared to conventional manufacturing.
Where there was an interest for us which immediately popped was for face shields. There are requests for these kinds of screens emerging all over the world, they are complementary protection elements that can help. The advantage of having a face shield is that it can easily be cleaned or disinfected, so it can be reused, and it allows for a certain degree of airflow without pain for the person who wears it.

C.S.: If any company can print these screens from the open-source files you made available, what about quality control and certification requirements?
Eric Bredin: These are questions that we are familiar with. So we wanted to stay in a field of application where the constraints are a little less complex than for medical devices such as ventilators. If we want to produce sterilizable face shields that can be reused, it means that we need to have the content, files, and materials to meet this demand. You have to be aware that in emergency situations the demands in terms of certification may not necessarily be the same, but people are saying “It’s better to have this than nothing.” Nevertheless, we are here with things we know well, we understand how it works, and by saying it can be done with this type of machine and this type of material, it allows us to offer a certain guarantee. If we decide to open up to other more complex devices, we can rethink the issues in a different way and review the notions of certifications.
C.S.: So far do you plan to do the same for more complex medical devices?
Eric Bredin: On our website, we try to collect information to find out what the needs are. Then with our technical, application and medical experts, we take a very pragmatic look. We analyze the answers to validate and say if it is technically possible, legally possible, possible from a safety point of view, and so on. We have to lock it all down before deciding whether or not to go ahead with it. For the moment, no decision has been made.
If we decided to go into applications that are elements or sub-elements of a more complete block, such as respirators, we couldn’t do so completely independently without validation through specialized companies, but we have technology and materials that can support biocompatible requirements.
C.S.: How many face shields will be produced? Are staff on-site and wearing masks?
Eric Bredin: Stratasys has started production in the United States. From an initial target of 5,000 masks during the first week, we expect to ship 120,000 by the end of April. It will depend on which hospitals are requesting them and which companies have the capacity to produce them, and then on our ability to assemble and supply the products.
With respect to personnel, there are waiver rules that allow certain staff to go to work to help companies produce masks. We have our medical experts who are familiar with the industry and who know the processes that need to be in place to ensure that production is done safely. We are not involved in the production of high-risk parts in relation to issues of exposure or transmission of the virus.
C.S.: What do you think of companies such as Airbus or Tesla that are embarking on 3D printing of complex devices such as respirators? Is this realistic? What about certification?
Eric Bredin: It’s not completely unrealistic. Of course, each business has its own peculiarities and complexities. If you talk to applied engineers who work in aeronautics, they are masters of their trade, masters of additive manufacturing. They know the challenges and how to manage mechanical, physical and chemical problems as complex as those found in the medical field: problems of flow, capacity, sealing, etc. So nothing is impossible at this level. Then it’s a matter of working closely with people who are more professionally oriented in the medical field and who can indeed bring their own recommendations and see how they can help continue to find solutions. I don’t see that as being inappropriate. These are good initiatives, they should be encouraged, especially if they are well integrated companies that are familiar with the processes and technologies.
As far as FDA certifications are concerned, I think that they are well versed in this, they may not have specific knowledge of the standard or the regulations in this area, but they know that it exists and it is not unfamiliar language to them. They know that in the medical field there are constraints, and that’s where they can work with experts like Medtronic who know the regulations, the control processes, etc. to get support at that level of detail.

C.S.: This type of provision of open source files with 3D printing instructions is growing. Is this solidarity specific to the COVID-19 situation?
Eric Bredin: The Coronavirus is an accelerator and it’s an eye-opener. All of a sudden everybody wants to be part of this goodwill momentum to try to bring quick solutions with 3D printing and to bridge the gap until traditional manufacturers take over and bring in the larger volumes of devices that hospitals need. But to use additive manufacturing before conventional manufacturing takes over is not actually a new way of working; it’s just more in the spotlight with the Coronavirus.
This is the concept of our GrabCAD community, a company acquired by Stratasys a few years ago. It is a community of designers and engineers who create files and exchange them. There is also GrabCAD print, which is related to software development and allows people who do design to go and transfer a file to a 3D printing machine. GrabCAD Shop is an element that allows someone who manages production to be able to deal with order and production management through a fleet of printers directly from his computer. These can be available within his own company, or from subsidiaries of companies, or even from external service providers. This fits very well with the notion of distributed manufacturing. Why not develop a GrabCAD shop for COVID-19 and try to give companies the opportunity to participate in the collective effort. In theory it is feasible, we have the tools and the technology, but in practice we have to mesh all this together. It is already happening for our COVID-19 initiative in the US.
C.S.: What are the advantages of additive manufacturing compared to conventional manufacturing?
Eric Bredin: First, the reactivity and speed of implementation. The time it takes to set up a 3D printer is the time it takes to download a file and send it to the printer. This speed is unbeatable. With conventional manufacturing methods, the implementation will be much longer and may take up to three weeks, the time to make the molds, etc. However, with conventional manufacturing, the manufacturing speed will be much faster once set up. 3D printing is still a technology that is not highly productive. For example, to manufacture a face screen, we’re talking about twenty masks produced per day on one printer. If there are a lot of printers, we can multiply the number, but in three days we will produce hundreds of masks, no more. But with a farm of printers like recently installed at AP-HP in Paris you manage thousands of parts per week.
Second, the ability to work in distributed manufacturing. Conventional methods with a centralized manufacturing center are certainly capable of addressing large volumes, but the problem is when the manufacturing center is decentralized. If overnight there is a blockage of air, sea or other transportation methods, you are stuck. So the advantage of additive manufacturing is decentralizing production by distributing files rather than manufacturing molds. You can send the files over connected distributed manufacturing networks.
Last is the flexibility of 3D printing. The process is not fixed. Once you’ve made a conventional mold, you can’t change it easily, whereas with additive manufacturing you have the advantage of being able to say at any time “I want to vary a small item,” for example if you want a face screen with a slightly larger head contour to adapt it to certain territories, it’s possible. You can easily customize some face shields.









